Bn molecular orbital diagram
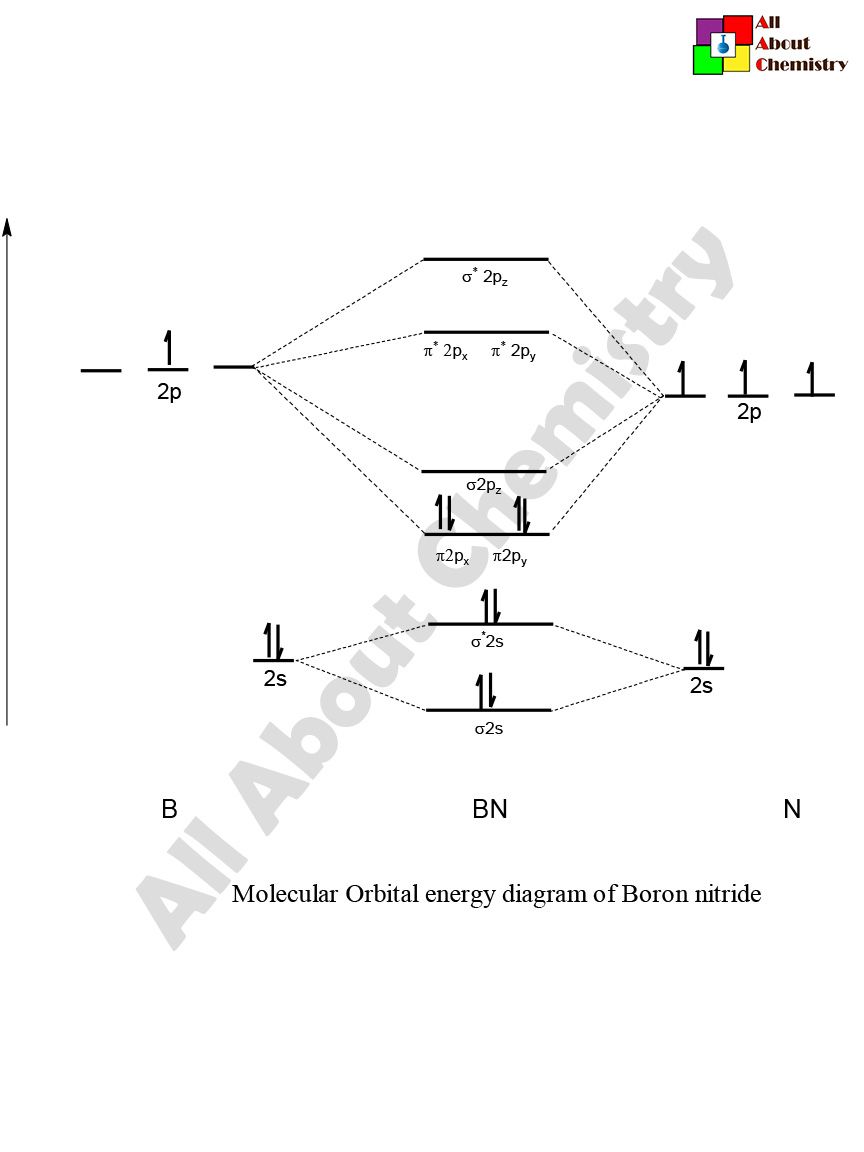
This is the general MO diagram you need to fill with the valence electrons of Bn molecular orbital diagram. Boron has 3 valence electronsand nitrogen has 5 valence electrons, this makes 8 electrons. You have to start filling the orbitals from those with lowest energy to those with higher energy. In this case, you need to follow Hund's rule, bn molecular orbital diagram, which states that if two or more orbitals of equal energy are available, electrons will occupy them singly before filling them in pairs.
None of the approaches we have described so far can adequately explain why some compounds are colored and others are not, why some substances with unpaired electrons are stable, and why others are effective semiconductors. These approaches also cannot describe the nature of resonance. Such limitations led to the development of a new approach to bonding in which electrons are not viewed as being localized between the nuclei of bonded atoms but are instead delocalized throughout the entire molecule. Just as with the valence bond theory, the approach we are about to discuss is based on a quantum mechanical model. In Chapter 2 , we described the electrons in isolated atoms as having certain spatial distributions, called orbitals , each with a particular orbital energy.
Bn molecular orbital diagram
Skip to main content. Table of contents. Intro to General Chemistry 3h 53m. Classification of Matter. Chemical Properties. Physical Properties. Intensive vs. Extensive Properties. Scientific Notation. Metric Prefixes.
Periodic Table: Classifications. Why are antibonding molecular orbitals higher in energy than the parent atomic orbitals?
.
This is the general MO diagram you need to fill with the valence electrons of BN. Boron has 3 valence electrons , and nitrogen has 5 valence electrons, this makes 8 electrons. You have to start filling the orbitals from those with lowest energy to those with higher energy. In this case, you need to follow Hund's rule, which states that if two or more orbitals of equal energy are available, electrons will occupy them singly before filling them in pairs. So you end up with 2 unpaired electrons, and paramagnetism of the molecule is explained. How to draw a BN molecular orbital diagram? Dec 11, This is the general MO diagram you need to fill with the valence electrons of BN Boron has 3 valence electrons , and nitrogen has 5 valence electrons, this makes 8 electrons.
Bn molecular orbital diagram
None of the approaches we have described so far can adequately explain why some compounds are colored and others are not, why some substances with unpaired electrons are stable, and why others are effective semiconductors. These approaches also cannot describe the nature of resonance. Such limitations led to the development of a new approach to bonding in which electrons are not viewed as being localized between the nuclei of bonded atoms but are instead delocalized throughout the entire molecule. Just as with the valence bond theory, the approach we are about to discuss is based on a quantum mechanical model. In Chapter 2 , we described the electrons in isolated atoms as having certain spatial distributions, called orbitals , each with a particular orbital energy. Just as the positions and energies of electrons in atoms can be described in terms of atomic orbitals AOs , the positions and energies of electrons in molecules can be described in terms of molecular orbitals MOs A particular spatial distribution of electrons in a molecule that is associated with a particular orbital energy. As the name suggests, molecular orbitals are not localized on a single atom but extend over the entire molecule. Consequently, the molecular orbital approach, called molecular orbital theory A delocalized bonding model in which molecular orbitals are created from the linear combination of atomic orbitals LCAOs , is a delocalized approach to bonding. Molecular orbital theory is a delocalized bonding approach that explains the colors of compounds, their stability, and resonance.
Reaper tattoo
In molecular orbital theory, electrons in antibonding orbitals effectively cancel the stabilization resulting from electrons in bonding orbitals. Power and Root Functions -. To determine what type of bonding the molecular orbital approach predicts F 2 to have, we must calculate the bond order. Double and triple bonds contain four or six electrons, respectively, and correspond to bond orders of 2 and 3. The diatomic molecule BN has never been detected. Introduction to Molecular Orbital Theory. Although the molecular orbital theory is computationally demanding, the principles on which it is based are similar to those we used to determine electron configurations for atoms. Sketch a molecular orbital diagram for BN. Writing Ionic Compounds. Instead, they are perpendicular to the internuclear axis. Filtration and Evaporation. Consequently, the electrons in the bonding orbitals are not shared equally between the two atoms. Conceptual Problems What is the distinction between an atomic orbital and a molecular orbital? Stoichiometric Rate Calculations.
None of the approaches we have described so far can adequately explain why some compounds are colored and others are not, why some substances with unpaired electrons are stable, and why others are effective semiconductors. These approaches also cannot describe the nature of resonance. Such limitations led to the development of a new approach to bonding in which electrons are not viewed as being localized between the nuclei of bonded atoms but are instead delocalized throughout the entire molecule.
Calculating Molar Mass. Paramagnetism and Diamagnetism. What is meant by the law of conservation of orbitals? Periodic Trend: Successive Ionization Energies. None of the other bonding models can predict the presence of two unpaired electrons in O 2. BN and C 2 are isoelectronic, with 12 valence electrons, while N 2 and CO are isoelectronic, with 14 valence electrons. Oxides, Peroxides, and Superoxides. If the highest occupied molecular orbital in both molecules is bonding, how will a one-electron oxidation affect the bond length? Ester Reactions: Esterification. Nonbonding Molecular Orbitals Molecular orbital theory is also able to explain the presence of lone pairs of electrons. We now turn to a molecular orbital description of the bonding in O 2.

Quite right! I think, what is it good thought. And it has a right to a life.
Many thanks for an explanation, now I will know.