Lewis dot diagram for mg
This chapter will explore yet another shorthand method of representing the valence electrons. The method explored in this lesson will be a visual representation of lewis dot diagram for mg valence electrons. We will, as we observed in the previous lesson, finish the lesson with a look at how this visual representation flows in a pattern throughout the Periodic Table.
File Talk. Read View on Commons. Tools Tools. This is a file from the Wikimedia Commons. Information from its description page there is shown below. Commons is a freely licensed media file repository.
Lewis dot diagram for mg
.
The same pattern holds for all elements in the main group.
.
In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. A Lewis electron dot diagram or electron dot diagram, or a Lewis diagram, or a Lewis structure is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The number of dots equals the number of valence electrons in the atom. These dots are arranged to the right and left and above and below the symbol, with no more than two dots on a side.
Lewis dot diagram for mg
In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. A Lewis electron dot diagram or electron dot diagram or a Lewis diagram or a Lewis structure is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The number of dots equals the number of valence electrons in the atom. These dots are arranged to the right and left and above and below the symbol, with no more than two dots on a side. It does not matter what order the positions are used. For example, the Lewis electron dot diagram for hydrogen is simply:. Because the side is not important, the Lewis electron dot diagram could also be drawn as follows:. By putting the two electrons together on the same side, we emphasize the fact that these two electrons are both in the 1 s subshell; this is the common convention we will adopt, although there will be exceptions later.
Hot emo dudes
You can help. The first and second valence electrons are placed on the side representing the s orbital and the next electrons are placed in the p orbitals. The method explored in this lesson will be a visual representation of the valence electrons. Policies and guidelines Contact us. These visual representations were given the name Lewis electron dot diagrams. To draw a Lewis electron dot diagram for sodium you can picture the symbol for sodium in a box with the box having four sides. This chapter will explore yet another shorthand method of representing the valence electrons. This is a file from the Wikimedia Commons. Summary Description Lewis dot Mg. Think of the chlorine in a box and the box has 4 sides.
This chapter will explore yet another shorthand method of representing the valence electrons. The method explored in this lesson will be a visual representation of the valence electrons.
File Talk. This chapter will explore yet another shorthand method of representing the valence electrons. One way to represent this valence electron, visually, was developed by Gilbert N. Read View on Commons. English: Lewis dot diagram for magnesium. We will, as we observed in the previous lesson, finish the lesson with a look at how this visual representation flows in a pattern throughout the Periodic Table. This file is made available under the Creative Commons CC0 1. Then we place the valence electrons around the sides of the box with each side representing an orbital in the outermost energy level. Sample Question. Items portrayed in this file depicts. The Lewis electron dot formula is: b Sulfur has the electron configuration: 1 s 2 2 s 2 2 p 6 3 s 2 3 p 4 , therefore there are 10 core electrons and 6 valence electrons.

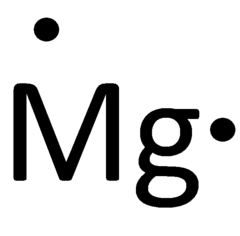
0 thoughts on “Lewis dot diagram for mg”