Lewis h2so3
The Sulfur atom has one lone pair while all the Oxygen atoms have 2 lone pairs. Note: Take a pen and paper with you and try to draw this lewis structure along with me, lewis h2so3. I am sure you will definitely learn how to draw lewis h2so3 structure of H2SO3.
In order to find the total valence electrons in H2SO3 molecule , first of all you should know the valence electrons present in hydrogen atom, sulfur atom as well as oxygen atom. Valence electrons are the electrons that are present in the outermost orbit of any atom. Hydrogen is group 1 element on the periodic table. You can see that only 1 valence electron is present in the hydrogen atom as shown in the above image. Sulfur is a group 16 element on the periodic table. Oxygen is group 16 element on the periodic table.
Lewis h2so3
Also, there is one lone pairs on sulfur atom. Concept of number of total valence electrons of atoms are used to draw lewis structure of H 2 SO 3. Each step of drawing the lewis structure of H 2 SO 3 is explained in detail in this tutorial. Sulfur atom is the center atom in H 2 SO 3 molecule. Three oxygen atoms are located around the sulfur atom. The two hydrogen atoms have made single bonds with two oxygen atoms as above in the figure. There are several steps to draw the lewis structure of H 2 SO 3. Those steps are explained in detail in this tutorial. Because H 2 SO 3 molecule is a bit complex molecule, almost all steps may be used. Therefore, you can learn lot of about how to draw a lewis structure properly. There are three elements elements in H 2 SO 3 ; hydrogen, oxygen and sulfur. Hydrogen is a group IA element and has only one electron in its last shell valence shell.
There are several steps to draw the lewis structure of H 2 SO 3.
.
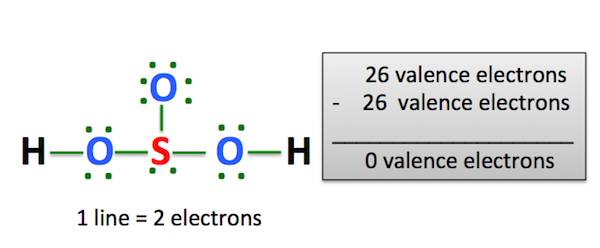
The key to understanding this Lewis structure is recognizing these two H's in front attached to a polyatomic ion. That makes it an acid. And these Oxygens here, the Hydrogens will attach to the outside of the Oxygens. So we'll put our Sulfur here in the middle, it's the least electronegative. We have three Oxygens. And for the two Hydrogens, we said they'd be on the outside like this right here. We have a total of 26 valence electrons for the H2SO4 Lewis structure. We'll put 2 electrons between atoms to form the chemical bonds there. We've used 10 valence electrons.
Lewis h2so3
H 2 SO 3 sulfurous acid has two hydrogen atoms, one sulfur atom, and three oxygen atoms. In the H 2 SO 3 Lewis structure, there are two single bonds and one double bond around the sulfur atom, with three oxygen atoms attached to it. The oxygen atom with a double bond has two lone pairs, the left oxygen and right oxygen atom with which the hydrogen atom is attached also has two lone pairs, and the sulfur atom has one lone pair.
Difference between soccer cleats and football cleats
Amphoteric nature of water NO 2 - lewis structure N 2 O lewis structure, resonance structures Stability of water. So it fulfills the octet rule. After deciding the center atom and sketch of H 2 SO 3 molecule, we can start to mark lone pairs on atoms. Also, only 24 valence electrons of H2SO3 molecule are used in the above structure. Concept of number of total valence electrons of atoms are used to draw lewis structure of H 2 SO 3. Oxygen is also a group 16 element on the periodic table. Now, you can see the electronegativity values of sulfur atom S and oxygen atom O in the above periodic table. H 2 SO 3 forms SO 3 2- ion which is an unstable ion in water. Sulfur is a group 16 element on the periodic table. In order to check the stability of the central sulfur S atom, we have to check whether it is forming an octet or not. Maximum valence of sulfur is six. I am sure you will definitely learn how to draw lewis structure of H2SO3. Total electron pairs are determined by dividing the number total valence electrons by two.
Attempts to concentrate the solutions of sulfurous acid simply reverses the equilibrium, producing sulfur dioxide and water vapor. Sulfurous acid is commonly known to not exist in its free state, and due to this, it is stated in textbooks that it cannot be isolated in the water-free form. Sulfurous acid is an intermediate species in the formation of acid rain from sulfur dioxide.
In order to find the total valence electrons in H2SO3 molecule , first of all you should know the valence electrons present in hydrogen atom, sulfur atom as well as oxygen atom. Now you have come to the final step in which you have to check the stability of lewis structure of H2SO3. Sulfur is a group 16 element on the periodic table. There is a one double bond between sulfur atom and oxygen atom. So you have seen the above image by now, right? Now, in the above structure, you can see that the charges are minimized and the above lewis structure of H2SO3 is the final stable structure. Valence electrons are the electrons that are present in the outermost orbit of any atom. Hydrogen's only valence is one and oxygen's maximum valence is two. Sulfur is a group 16 element on the periodic table. Those steps are explained in detail in this tutorial.

I consider, that you commit an error. I can prove it. Write to me in PM, we will discuss.
Bravo, what necessary phrase..., a brilliant idea
Curious question