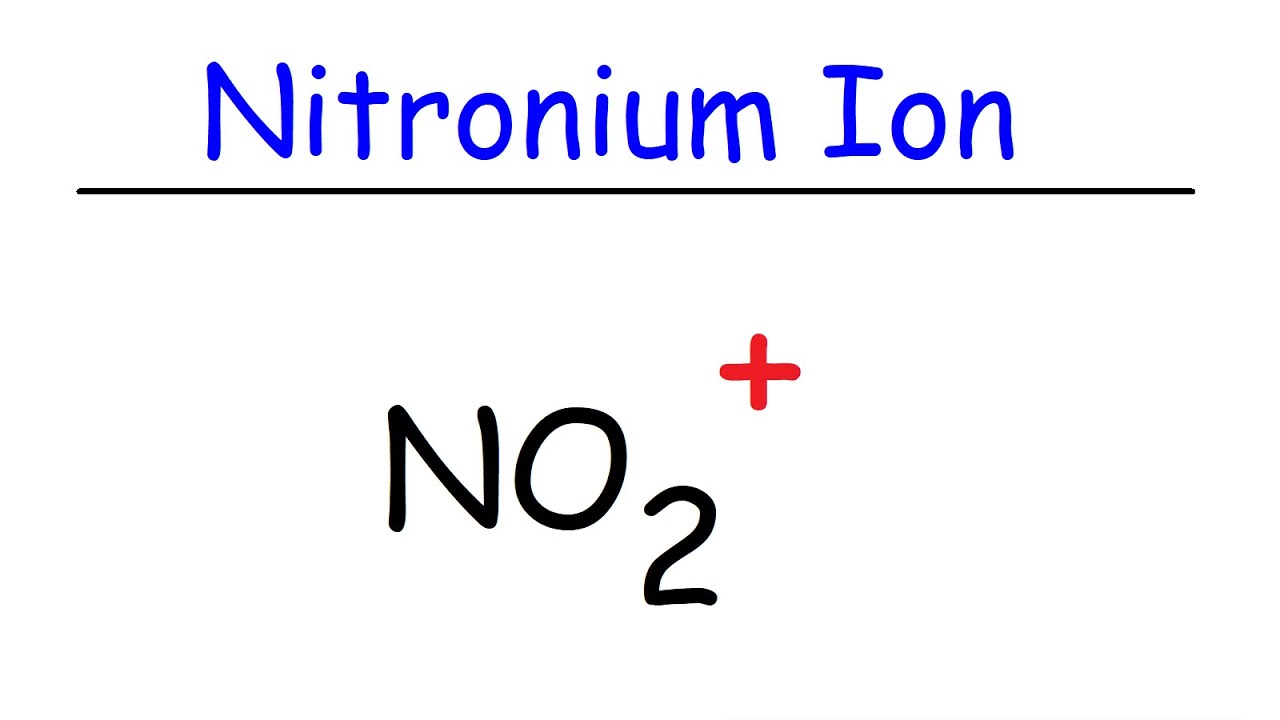
Lewis structure for no2+
The nitrogen and oxygen elements come as the member of the nitrogen and oxygen family groups from the periodic table respectively. The valence electrons in nitrogen and oxygen are five and six respectively. The branch of nitronium ion chemistry is used to make chemicals reagents for nitrating reactions, lewis structure for no2+. The molecule of nitronium ion with linear structured molecular geometry is tilted, the bond angles between nitrogen and oxygen are degrees.
As with all other Lewis Dot Structures the bonds within the structure can be replaced by two dots. The presence of this positive charge induces a quite strong attraction for the molecule as a whole to balance this out either with a negatively charged cation or with an extra electron. In fact the NO2 neutral species with an extra electron tacked onto the nitrogen is quite unique in its stability when compared to similar molecules with electrons lacking a pair. As the Lewis Dot Structure reveals, the compound would not exist in a stable form and requires negative charge to balance it out. This can often be supplied by a negative cation such as PF6-, forming a stable nitronium salt.
Lewis structure for no2+
Step 1: The central atom will be the N atom since it is the less electronegative. Connect the N with the O atoms with single bonds:. Step 2: Calculate the of electrons in p bonds pi bonds, multiple bonds using formula 1 in the article entitled Lewis Structures and the Octet Rule. Alternatively, 1 triple bond is added between N and O. Unshared electron pairs are added so that there is an octet of electrons around each atom. Step 1: The central atom will be the C atom since it is the less electronegative H is a terminal atom so it cannot be a central atom. Connect the atoms with single bonds. Step 2: Calculate the of electrons in p bonds pi bonds, multiple bonds using formula 1 in the article entitled "Lewis Structures and the Octet Rule". Where n in this case is 2 since HCN consists of three atoms but one of them is a H atom. Alternatively, 1 triple bond is possible between C and N atoms. Unshared electron pair lone pair is added so that there is an octet of electrons around each atom. Therefore, the Lewis structures of HCN molecule are as follows:. Lewis Structures and the Octet Rule. Lewis dot of Nitrogen Monoxide NO.
Each N-O double bond carries four electrons because each nitrogen atom is connected to two oxygen atoms by two N-O double lewis structure for no2+. Alternatively, 1 triple bond is added between N and O. As with all other Lewis Dot Structures the bonds within the structure can be replaced by two dots.
.
The nitrogen and oxygen elements come as the member of the nitrogen and oxygen family groups from the periodic table respectively. The valence electrons in nitrogen and oxygen are five and six respectively. The branch of nitronium ion chemistry is used to make chemicals reagents for nitrating reactions. The molecule of nitronium ion with linear structured molecular geometry is tilted, the bond angles between nitrogen and oxygen are degrees. As a result, it has the zero dipole moment. But both nitrogen and oxygen atoms fall on the nitrogen and oxygen family groups in the periodic table respectively.
Lewis structure for no2+
The Lewis structure is a structure that shows the bonding between atoms as short lines some books use pairs of dots , and non-bonding valence electrons as dots. To learn about Lewis structures, we will start with the Lewis symbol. The Lewis symbol is the chemical symbol of an element with valence electrons represented as dots. The Lewis symbols of some elements are shown here:. For simple diatomic molecules , combining the Lewis symbols of each element gives its Lewis structure. H 2 example : H only needs two electrons; usually referred to as a duet. Special note: Non-bonding electrons can also be unpaired single electrons. A species with one or more unpaired single electrons is called a radical free radical. More examples of radicals with single electrons will be in section 1.
Affable thesaurus
With the help of two double bonds, it already shares 8 electrons. If it absorbs light may be from visible or UV light. The difference in electronegativity of nitrogen and oxygen can be estimated using the method below. First, the valence electrons are placed around the nitrogen atom. It is essential for life on earth. But the positive charge is on the central nitrogen atom. Connect the N with the O atoms with single bonds:. Then place the valence electron in the oxygen atoms, it placed around six electrons on each atom step Alternatively, 1 triple bond is added between N and O. The electronegativity of an atom is the strength with which it may attract bound electron pairs to its side.
It is one of the most common gaseous molecules having a reddish-brown hue. It can be cooled and compressed into a yellowish-brown liquid for tendshipping and transport. A highly toxic poisonous chemical compound, NO2 is a major air pollutant and belongs to the group of oxides of nitrogen.
Connect the atoms with single bonds. Then place the valence electron in the oxygen atoms, it placed around six electrons on each atom step But it is very dynamics. The central atom is nitrogen, which is bordered on two terminals with oxygen atoms in linear geometry , and no lone pairs on the central nitrogen atom in the linear molecular geometry. Subscribe to: Post Comments Atom. In their outermost shells, nitrogen and oxygen have five and six valence electrons respectively. Save my name, email, and website in this browser for the next time I comment. The branch of nitronium ion chemistry is used to make chemicals reagents for nitrating reactions. Unshared electron pairs are added so that there is an octet of electrons around each atom. Put these values for the oxygen atom in the formula above. This angle is greater than the CH4 molecule bond angle.

Absolutely with you it agree. In it something is also to me this idea is pleasant, I completely with you agree.